
Meet Scientists, Industrialists, Professors, Researchers from UAE, Europe, USA, Middle-east, Asia Pacific, UAE, Dubai at Green Catalysis Congress, Sustainable Energy conferences Green Engineering events, Chemical Engineering Meeting 2018, 2019
Green Catalysis Conference | Sustainable Energy Congress| Green chemistry Meetings | Chemical Engineering Events | Biocatalysis Conference | Green Nanocatalysis | Asia Pacific | Dubai | USA | Europe | Middle East | UK | Canada
Theme: Promoting Sustainability through Green Chemistry!!
Green Catalysis 2018
- About Green and Sustainable Energy Conference
- Sessions/Tracks
- Market Analysis
- New Updates- Green Catalysis 2018
Green Catalysis 2018 Conference
Green Catalysis 2018 invites all the scholars, researchers from all over the world to attend and present their respective scientific research at “Annual Conference on Green Catalysis and Sustainable Energy” held November 6-7, 2018 at Dubai, UAE which includes prompt Keynote presentations, Oral talks, Poster presentations, Delegate views, and Exhibitions.
Green Catalysis 2018 conference is all about providing a resourceful and knowledgeable ground for the associates in the field of Green Catalysis Research, Green Chemistry Application, and Sustainable resources in the latest generation. As the theme of the conference relates it serves as a global platform to converse concerning the current developments, recent advances, new approaches and future approaches in the field of Chemistry Research. Attempts are to provide a perfect stage to share knowledge and experiences and encourage people to carry out effective researches and works to combat the Chemistry Research & its application. It is all about Inspiring and get Inspired!! We have research expertise from different parts of the world coming to share their latest research works in the field of Chemistry Research, Chemical Engineering science. And to inspire the young minds we are glad to present a special category - The Young Researcher Forum (YRF).
Green Catalysis 2018 Conference purpose is to fill your head with the knowledge you can use: ideas, new trends, amazing ingenuity. Our focus is on Sustainable Development and Green Technologies, which we believe are foundational to the success of individual organizations as well as our cities, states, nations, and world. Attendees come to Green Catalysis Conference to learn from experts in their community and leave.
Target audience:
Target Audience will be personnel from both industrial and academic fields which include;
- Green Chemists
- Chemistry Associations
- Chemical Industries
- Associate and Assistant Professors in Green Chemistry
- Pharmaceutical Professionals
- Industrial Experts
- Industry Safety Officers
- CEOs, COOs, Directors, Vice Presidents, Co-directors, Managing Directors,
- Post-Doctoral and Researchers in Chemistry, Students from the related fields.
Highlights of latest advances in Green Catalysis
Track 1: Green chemistry and catalysis
Green chemistry otherwise called as sustainable chemistry, which is part of chemistry and chemical engineering focused on designing products and by minimizing the generation and use of hazardous substances .whereas, environmental chemistry focuses on the effects chemicals polluting the nature, green chemistry focuses on technological ways to prevent pollution and by reducing the consumption of non-renewable resources.
Catalysis is the defined as the substance which only alters the rate of the reaction without changing is nature. In order to achieve objectives of the Green chemistry, catalysis plays a fundamental role. The main goal of green chemistry is to provide an eco-friendly, reusable, recyclable and minimum waste production and green catalysis design the chemical products in a way that reduces or eliminates the use and generation of hazardous substances.
- Homogenous and Heterogeneous Green catalysis
- Application of green chemistry in Catalysis
- Green chemistry and Organocatalysis
- Biocatalysis
- Green Chemistry catalysis and sustainable energy development.
Track 2: Bio gas and Bio Fuel In Sustainable Energy
The expensive cost of producing biogas, biopropanol and biobutanol limits their use; bioethanol is produced and used widely in Brazil while some countries favour the use of biodiesel. Bioethanol is produced by fermentation from corn (maize), cassava, sugar cane, sugar beet and various lignocelluloses biomass. Hydrolytic conversion of carbohydrates (hemicelluloses and cellulose) in plants is a route to bioethanol production; this is a yeast catalyzed anaerobic conversion or digestion of sugars to alcohol. Anaerobic hydrolysis of biological wastes results in the production of biogas which is a mixture of methane and carbon dioxide . Bioethanol is a promising biofuel that can be derived from any material containing simple or complex sugars. It can be produced from sugar cane and starchy foods such as corn, wheat, potatoes. Lignocellulose has been described as the most promising raw material, cellulose is the most common biopolymer present in wood, organic industrial wastes and is a polysaccharide that can be converted into sugars and fermented. Thus, bioethanol from lignocellulosic materials has the potential to be a valuable substitute for, or complement to, gasoline The polysaccharides cellulose and hemicellulose in lignocellulose, are not readily available
- Reduction in sources
- Incorporate early Sustainability in the design process
- To Create Industrial Processes that Avoid Hazard Problems
- Development of environmentally friendly chemicals and materials
- Analysis of the eco-toxicological and environmental effects of biomass processing
- Use of the environmentally organic Solvent system
- Regenerating of Waste materials
Track 3: Sustainable Energy in Water Resources
Sustainable water management is part of sustainable development; meeting the needs of the present without compromising the ability of future generations to meet their own water needs. Achieving sustainable water management requires a multidisciplinary and holistic approach in which technical, environmental, economic, landscape aesthetic, societal and cultural issues are addressed. Further research is required to guide the development of appropriate sustainable water management measures, strategies and policies.
- Wastewater Treatment
- Global Change
- Stormwater Management
Track 4: Polymer Engineering and Nanotechnology
Polymer designing is a sub-field of materials building basically concentrating on the improvement of new items. Polymer designs frequently ponder plastics, albeit different substances are likewise viewed as polymers .With preparing in polymer building and innovation, you may function as a polymer build, examining plastics and different polymers at the atomic level, choosing polymers for new applications and testing or handling plastics for new items. On the other hand, you may pick not so much logical but rather more generation based work as a building professional or technologist, concentrating on separating wood, cotton, petrochemicals and other crude materials to make polymers in a mechanical plant.
- Green Nanotechnology Synthesis
- Immerging materials for Green Catalysis
- Organocatalysis: Key Trends in Green Synthetic Chemistry
- Cleaning Up Oil Spills
- Solar cells
- Optoelectronics
- Graphene Devices
- Nano devices
- Nano remediation and water treatment
- Environmental remediation
- Nanofiltration
Track 5: Renewable Energy & Climate Change
The falling expense of sustainable power source technologies , notably sunlight based and wind control, contributes extensively to the developing intensity of renewables opposite customary energizes. Sun oriented PV modules, for example, cost seventy five percent less today than in 2009, while wind turbine costs have declined by right around a third finished the same period. Cost decreases combined with viable empowering arrangements have implied that sustainable power source limit increments have kept on outpacing those of atomic and non-renewable energy sources in the power division since 2011. For this amazing development to end up worldwide, encourage speculation is required in nations and areas that are setting out on a change of their vitality systemsover the coming decade.
- Batteries and Vehicles
- Choice of catalysts
- Choice of reagents
- Renewable sources
Track 6: Nanotechnology in Oil and gas Application
The light vaporous hydrocarbons created by synergist breaking are exceptionally unsaturated and are normally changed over into high-octane gas segments in polymerization or alkylation forms. In polymerization, the light olefins propylene and butylene are prompted to consolidate, or polymerize, into particles of a few times their unique atomic weight. The impetuses utilized comprise of phosphoric corrosive on pellets of kieselguhr, a permeable sedimentary shake. High weights, on the request of 30 to 75 bars (3 to 7.5 MPa), or 400 to 1,100 psi, are required at temperatures running from 175 to 230 °C (350 to 450 °F). Polymer fuels got from propylene and butylene have octane numbers over 90. The alkylation response additionally accomplishes a more drawn out chain particle by the mix of two littler atoms, one being an olefin and the other an iso paraffin (normally isobutane). Amid World War II, alkylation turned into the principle procedure for the fabricate of isooctane, an essential part in the mixing of avionics fuel. Two alkylation forms utilized in the business depend on various corrosive frameworks as impetuses. In sulfuric corrosive alkylation, concentrated sulfuric corrosive of 98 percent immaculateness fills in as the impetus for a response that is completed at 2 to 7 °C (35 to 45 °F). Refrigeration is vital as a result of the warmth created by the response. The octane quantities of the alkylates delivered run from 85 to 98
- Chemoselectivity
- Regioselectivity and diastereoselectivity
- Enantioselectivity
Track 7: Green chemistry and Polymer technology
Today polymer technology plays a significant role in the society as the application of polymer products has become much important in our life. Issues such as extensive use of the non-renewable source of energy, application of reagents that are a concern to the environment, generation of waste and many other make scientists and researcher think for new innovation which is sustainable and environmental friendly in the polymerization process. The utilization of renewable energy and green chemistry techniques has enhanced the new successful ways in polymer technology. Biodegradability of polymer materials is an auxiliary benefit of renewable polymers. Polymer manufactured by the biological system is essential biodegradable. Polymer science is not only applied for manufacturing a different variety of products but also it is utilized in the generation and saving of energy and at the same time it is improving renewable energy technology.
- Green chemistry and polymer synthesis
- Green and sustainable polymers
- Biopolymers
- Bioactive and biohybrid polymers
- Green and sustainable polymers
- Green composite polymer
- Polymer membranes for biomedical and separation techniques
- Green Polymer technology and waste management
- Polymers for energy generation and storage
- Polymeric solar cells
Track 8: Green Chemistry in Pharmaceuticals
The process provides an innovative way to traditional extractive procedures and highlights the increasing biotechnology applications in greener drug manufacturing. With improved process conditions and incorporating Green chemistry in the synthesis of active pharmaceutical ingredients is the importance of the pharmaceutical industry. Biocatalysis, solvent reduction and replacement are the same tools used in optimizing active pharmaceutical ingredient syntheses. With the applications of this technology synthetic oligonucleotide are emerging as a class of drug molecules with a broad spectrum in therapeutic application.
Track 9: Petrochemical Industries
Green chemistry and green catalysis principles play a key role in developing a number of chemicals but in an alternative way from petroleum and other non-renewable sources of energy. With growing advancement in the field of biotechnology, genetics, chemistry, and engineering leading to a new concept for converting renewable biomass into valuable products and fuels through innovations by producing bio-based chemicals, which is a result of a coupling reaction of chemical and biological products.
Track 10: Green catalysis and Pollution control
Pollution control is a process of reduction or elimination of waste at the source level by modifying the production process by using less or non-toxic substances, by implementing new conservation techniques and reusing of materials rather than releasing into water bodies or land. Pollution control is an environmental management which means the control of effluents and emission of wastes in water, air, and soil. By the application of green chemistry, the hazardous chemicals are eliminated at the molecular level.
Track 11: Green chemistry metrics
Green chemistry metrics measure the aspects of a chemical reaction in relation to Green chemistry and quantify the effect of the environmental performance of a chemical process which in turn allows the changes in performance to be measured.
- Effective mass yield
- Carbon efficiency
- Atom economy
- Reaction mass efficiency
- Environmental (E) factor
Track 12: Solar Cell and fuel cell
A fuelcell is one of the tools for converting the sustainable energy sources like solar radiation, wind, and biomass into a usable form of energy. Fuel cells are the devices which convert chemical energy into one form or other because of the efficiency and ability to harvest energy from the diverse source. The fuel cells are one of the keys to sustainable power generation and are used in space shuttles to generate water and electricity required for the mission. Green catalysis and fuel cell technology offer the opportunity to eliminate expenses by providing electricity in an efficient manner and make use of waste by-products which is produced in a treatment process.
Track 13: Green chemistry catalysis and Sustainable energy
Application of Green chemistry and its application strongly support the development of greener concepts in process parameters, selection of compounds and resulting environmental aspects. Successful implementation of green chemistry research helps in analyzing of new and existing green chemistry technologies are improving the environmental impacts of chemical products and processes with qualitative and quantitative benefits to the environment.
Track 14: Green energy
Green energy also referred to as Renewable energy is the energy that is originated from natural sources such as the sun, water, wind rain, waves, tides, plants, geothermal heat and many more. The energy generated from these resources is Clean, Varied, Renewable, Stable, and Inexpensive, which makes it more effective and demanding. The energy from these resources is utilized readily for purposes such as generating electricity, water heating, solar cooling, and a variety of commercial and industrial uses. The development in renewable energy technology has increased the efficiency of energy production by the renewable resources and at the same time, it has also decreased the dependence on non-renewable sources like oil, gas, coal, and utility which gets depleted with use.
- Solar Energy
- Hydropower
- Wind Energy
- Geothermal Energy
- Ocean Energy
- Bioenergy
Track 15: Green economy
The green economy strives to use alternative methods to promote sustainability in the reduction of energy and demand of raw materials, to prevent environmental pollution, to reduce Greenhouse emission, minimize waste by conversion of industrial wastes by microbial actions and effective recycling of wastes and by-product. The global market for Green economy technology has grown for years. The fastest growing sector includes energy storage, transportation, energy generation, energy efficiency, recycling and waste treatment. The emphasis on green economic growth provides an opportunity for the advancement of Green chemistry approaches.
Track 16: Energy Management and Energy Sector
One of the primary mainstays of the cutting edge industry is the continuous supply of vitality with a sensible cost. The vitality generation depends for the most part on non-sustainable power source assets that are getting more costly continuously. This expansion in vitality cost thusly drives the organizations to a more costly generation of merchandise. In this way it has been set up that vitality effectiveness isn't just critical for the earth yet additionally for the supportable creation in the assembling organization.
- Energy Sales
- Energy Projrcts
- Commercials
- Energy Marketing
Aim and Scope
Catalyst is playing a vital role in most of the chemical reaction. Catalysis is the centre face of synthesis of any substance from academic research to industry. A variety of products such as medicine, chemicals, fuels, polymers and many more which are highly useful for human need will not be feasible in the absence of catalysts. Chemical industries widely use catalysis process. Chemical Industries has exploited the catalyst to produce their required products. And Traditional catalysis process has many demerits such as:
-
High cost
-
It causes pollution to the environment
-
Tough to separate from the product
-
Ruined by impurities
-
They poison the product some time
Green Chemistry and Catalysis
Green Chemistry provides an exceptional platform for the development of innovative research and work on the development of alternative sustainable technologies.
These have led to the development of environmentally friendly and economical chemical processes in chemical industries. Development in Green Chemistry has able to make the things easy. Green chemistry also called as sustainable chemistry can be defined as process of chemical science and technology which is utilized to manufacture different desired products with sustainable, safe, non-polluting, consumes minimum amount of materials and energy and at the same time producing very less or no waste. It reduces the use and generation of hazardous substances in the manufacturing or application of any chemical product.
Green chemistry is an interdisciplinary subject, which combines chemistry, chemical engineering, toxicology, and ecology. Development in Green Chemistry has led to the innovation of new catalysis process which has high performance, economically efficient, and minimum consumption of resources. This green catalyst Has High performance not only with Environmentally Prospective but also with economic efficiency and also minimum consumption of resources. The application catalysis has reduce toxicity.
Why Dubai?
Dubai is the most popular and largest city of United Arab Emirates (UAE). The geographic location covering the southeast coast of the Persian Gulf is the capital of the Emirate of the Dubai; the country is being all of seven emirates. Dubai is situated roughly at sea level 16m (52 ft. above) on the Persian Gulf coast of the United Arab Emirates. The Dubai Emirates shares its boarders with Abu Dhabi in the south, Sharjah in the northeast, and the Sultanate of Oman in the southeast. Dubai is positioned at 25.2697°N 55.3095°E and covers an area of 1,588 sq. mi, which represents a significant expansion beyond its initial 1,500 sq. mi designation due to land reclamation from the sea. Dubai being a tourism and business place attracting the tourist through the giant hotel Burj Khalifa, Dubai Creek, Artificial archipelago called Palm Jumeirah, and many more exciting places.
Green Catalyst market
Green Catalyst market was valued at $28,567 million, and is expected to reach $40,000 million by 2022, supported by a CAGR of 4.8%.
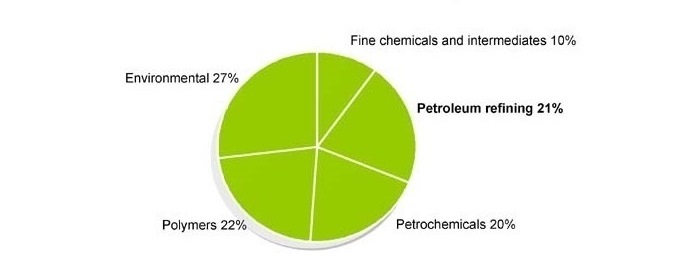
The graph above shows the application of Green Chemistry products in other Industries.
The size of the green chemical industry at 2015 was approximately $11.0 billion worldwide. It will likely grow about $100 billion in 2020.
Biocatalyst market
The global Biocatalysis market size was USD 8.18 billion in 2015 and is expected to witness significant growth over the next eight years on account of its increasing application in detergents, pharmaceuticals, and food & beverages.
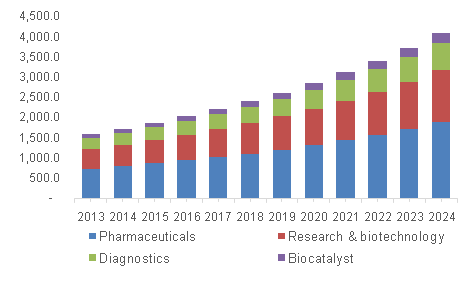
The Graph above shows the growth of increasing application of Biocatalysis in different other fields.
Related Conferences
1. 9th Annual Congress and Expo on Biofuels and Bioenergy, April 16-17, 2018 Dubai, UAE
2. International Conference on Polymerization Catalysis and Flexible Polymer, September 06-07, 2018 Dubai, UAE
3. 5th World Congress on, Catalysis and Chemical Engineering, September 5-6, 2018 Tokyo, Japan
4. 2nd Annual Congress on Environmental Pollution and Global Warming, June 21-22, 2018 Osaka, Japan
5. 5th World Congress on Green Chemistry and Green Engineering, July 19-20, 2018 Melbourne, Australia
6. 9th World Convention on Recycling and Waste Management, October 22-23, 2018; Osaka, Japan
7. 11th Global Experts Meeting on Chemistry And Computational Catalysis, May 18-19, 2018 Singapore
8. 11th World Bioenergy Congress and Expo, July 02-04, 2018 Berlin, Germany
9. 5th World Congress and Expo on Green Energy, June 14-16, 2018 London, UK
10. 4th International Conference on Pollution Control & Sustainable Environment, July 26-28, 2018 Rome, Italy
11. 4th International Conference on, Polymer Chemistry, June 25-27, 2018 Stockholm, Sweden
12. 9th World Congress on Green Chemistry and Technology, September 17-19, 2018 Amsterdam, Netherlands
13. 8th International Conference on Environmental Chemistry and Engineering, September 20-22, 2018 Berlin, Germany
14. 2nd International Conference on Renewable Energy and Resources, August 27-28, 2018, Boston, Massachusetts, USA
15. 36th International Conference on Environmental Chemistry & Water Resource Management, September 24-25, 2018 Chicago, Illinois, USA
16. International Conference on Catalysis and Pyrolysis 2018 (ICCP-2018), November 05-06, 2018 San Francisco, California, USA
17. 2nd International Conference on Nanostructured Materials & Nanochemistry, November 02-03, 2018 San Francisco | California | USA
18. 3rd International Green Catalysis Symposium, March 23 and 24, 2017, Rennes France
19. 20th International Conference on Heterogeneous Catalysis and Green Chemistry, January 15 -16, 2018, Zurich, Switzerland
20. 3rd Green & Sustainable Chemistry Conference, 13-16 May 2018, Berlin, Germany
21. 2nd International Conference on Catalysis and Chemical Engineering, February 19-21 2018, Paris, France
22. 5th International Conference on Green Chemistry and Technology, July 24-26 2018, Rome, Italy
23. International Conference on Green and Sustainable Chemistry, July 23-26, 2017, Melbourne, Australia
24. 2nd International Conference & Expo on Green Chemistry and Engineering, July 23-24, 2018, Barcelona, Spain.
25. Conference on Advances in Catalysis for Energy and Environment, January 10-12, 2018, Mumbai, India
26. International Symposium on Catalysis and Speciality Chemicals ISCSC 2018, October 1-3, 2018, Tlemcen, Algeria
27. 28th International Conference on Organometallic Chemistry (ICOMC-2018), July 15-20, 2018, Florence, Italy
28. 7th International Conference on Green Chemistry and Technology June 18, 2018, Dublin, Ireland
29. The 3rd International Conference on Green Chemistry and Technology, July 14-16, 2018, Kunming, Yunnan, China
30. 32nd EFFoST International Conference, 6-8 November 2018, Nantes, France
Related Associations and Societies
Middle East
- Chemical Society of Turkey
- Gulf Petrochemicals & Chemicals Association
- International Council of Chemical Association
- Kuwaiti Chemical Society
- Saudi Arabian International Chemical Sciences
- Renewable Energy Saudi Arabia
- Saudi Arabia Solar Industry Association
- Jordanian Chemical Society
- Iranian Chemists Association
- Iranian Nanotechnology Laboratory Network
- International Association of Catalysis Societies
- Iranian Biofuel Society
Asia pacific
- Chinese Chemical Society (Beijing)
- The Catalysis Society of India
- Chinese Chemical Society
- Chemical Research Society of India
- Chinese-American Chemical Society
- Indian Chemical Society
- Japan Society of Clinical Chemistry (JSCC)
- Siberian Branch of Russian Academy of Sciences
- Serbian Chemical Society
- Society of Chemical Industry
- Society for Applied Spectroscopy
- Indonesian Association for Clinical Chemistry
- Japan Association for International Chemical Information
- Federation of Asian Chemical Societies
- Hong Kong Society of Clinical Chemistry
- Institute of Chemistry
- CeySerbian Chemical Societylon
- Asia-Pacific EPR/ESR Society
- Nordic Catalysis Society
- Federation of Asian Chemical Societies
- Society of Environmental Toxicology and Chemistry (SETAC
- Bangladesh Chemical Society
- Bangladesh Atomic Energy Commission
- Competence Centre for Catalysis, Sweden
- The Korean Chemical Society
- Centre for Nano and Soft Matter Sciences
- Indian Association for the Cultivation of Science
- Iranian Nanotechnology Laboratory Network
- Materials Research Society
- Dutch Institute for Catalysis Research
Europe
- European Chemical Society
- Royal Society of Chemistry, United Kingdom
- The European Association of Catalysis Societies
- Society of Chemical Industry, European Union
- Hungarlian Chemical Society
- https://green-catalysis.conferenceseries.com/abstract-submission.php
- Italian Chemical Society
- International Network for Sustainable Energy
- Swedish Chemical Society
- Royal Netherlands Chemical Society
- Société Chimique de France
- RenewableUK
- Belgian Society of Biochemistry and Molecular Biology
- Swedish Chemical Society
- Federation of European Biochemical Societies
- Pancyprian Union of Chemists
- Institute of Chemistry of Ireland
- Association of Greek Chemists
- Gesellschaft für Chemische Technik und Biotechnologie, Germany
- Norwegian Chemical Society
- Serbian Chemical Society
- Society of Chemical Industry
- Bristol Centre for Functional Nanomaterials
- German Catalysis Society
- European Federation of Chemical Engineering
- Federation of European Zeolite Associations
- Royal Australian Chemical Institute
- The Catalysis Society of Australia
- Federation of African Societies of Chemistry
- Australian Institute for Bioengineering and Nanotechnology
- BioNano Laboratory, Canada
- Organic Reactions Catalysis Society
USA
- Biomass Energy Research Association
- Polymer processing society
- Michigan Green Chemistry Clearinghouse
- Green Technology Business Innovation & Trade
- International Union of Pure and Applied Chemistry, USA
- Canadian Society for Chemical Technology
- American Society for Mass Spectrometry
- American Institute of Chemists
- Nanomedicine Roadmap Initiative
- Nanotechnology Industries Association
- Catalysis Society of Japan
- Federation of Iberoamerican Catalysis Societies
- North American Catalysis Society
New challenges encountered during Membrane Separation Processes in Water Purification
The growing water pollution and climate change have critically affected the freshwater supplies. Human activities like discharging the chemicals into the ground have degraded the quality of water resources like groundwater and the declining of centralized water distribution systems has hampered the natural resources. These have forced us to the bring new techniques for the better conservation and management of existing resources and at the same time to make the non-traditional sources of water like seawater, brackish water, and wastewater to become a part of water supply. Membrane separation technique has emerged as a highly efficient technique for the desalination of seawater by reverse osmosis and membrane bioreactor process. Membrane separation technique has gained a lot of attention in today’s water treatment process due to the reason of its easy-to-operate, low energy demand, ability to produce water of high purity, and it’s simple to designs with small footprints.
Membrane separation processes are mainly classified into desalination processes or hybrid membrane processes. The process used to produce only potable water from a saline or wastewater source (i.e. single purpose) is referred to as desalination processes. Whereas membrane processes which include several processes for different purpose such as nutrient recovery, metal capture, harvesting energy and generating potable non-potable water for the purpose of some target applications.
Desalination process has been successful in large scale but it still faces some challenges that need to be correctified before its widespread application. Some of the major challenges faced in Desalination process are the management of the concentrate stream which is one of the most important concerns. There are some processes such disposing the brine again into the ocean, which still has the risk of desalination of inland sources (e.g., brackish water). Furthermore, this desalinating membrane faces material challenge i.e. these membrane materials are highly-selective in nature, they still permit small amounts of solutes such as boric and chlorine ions to permeate. This is the reason which does not permit the use of desalinated water for agricultural purposes. It can only be used after further processing of the desalinated water. Moreover, the brine contains micropollutants and other contaminants that are risks to the environment and human health. The disposal of this brine, however, is a challenge for the desalination process. Membrane fouling and Degradation by chlorine is one of the other common issues regarding Desalination process. Finally, Desalination forms are vitality concentrated on the grounds that they focus on the treatment of saline water sources, and the high osmotic weights produced by these sources require a lot of vitality to overcome
Nanocatalysis for Sustainable Energy Development
There are numerous difficulties for catalysis has made continue toward the path to analyze for new conceivable possibilities of nanocatalysis for sustainable energy development. Catalysts are redesigned in nano-dimensional forms which have shown have that has the capability to enhance their performance and increase their stability and lifespan. With the increasing demand of energy and as well as with the depletion of the non-renewable source of energy and at the same time with the significant concern of environmental pollutions, the designing, understanding, and development of nanocatalysts will play an important role in ensuring a sustainable future. Presently Green Catalysts with superior performance, selectivity, and lifespan for chemical processes and renewable energy resources is one of the most important research topics in the field of sustainable energy development. An exciting new range of possibilities are offered in terms of the design of nanocatalysts and to develop novel solutions for a sustainable energy future. With the development of advanced nanotechnology, the potential has also increased the potential of the catalyst such as novel nano carbons are highly utilized as catalysts for advanced energy conversion and storage devices. Nanocatalysis has also developed the production of solar fuel which has enabled the transition to a new sustainable energy system. Thus nanotechnology and catalysis strategies can solve the energy challenge and for a smarter use of renewable energy.
New development in Fuel Cell Technology
Increase in carbon emission has become a great concern in the modern scenario. This has led to the development of the fuel cell technology for achieving a carbon future. Development of fuel cell technology has led the generation of electricity from solid carbon like biomass and coal in more clean and efficient.
A fuel cell is classified into several types based on the type of electrolyte they use. Among all, Phosphoric Acid Fuel Cell has reached the commercial purpose. Gases from sewer digester and biogas formed by methane fermented organic raw material or industrial wastewater are used for PAFC which cost’s very less. PAFC has higher power generation efficiency and its cost is also economically advantageous. However further advances such as colt reduction enabled by further research and development expanded the use of biogas or sewer digester fuel a wider application such as supplying power to a critical load is expected to pave the PAFCs market. The polymer electrolyte Fuel cell was actively developed which was highly beneficial for power generation of smaller capacity then PEFCs and hence decreases the waste heat temperature level than PAFCs. PEFCs have low reaction temperature approximately 80 degree Celsius above room temperature and use inexpensive materials.
Food supply chain waste: Production of biodiesel from waste cooking oil.
There is a huge demand for fuels and chemicals in today scenario and the main factors behind this are overpopulation. There is a big threat of global warming and depletion of fossil fuel that force our resource system and thus these consequences make necessary for the development of sustainable and innovative strategies for the chemical industry. Currently, the world is experiencing the restriction imposed due to the limitation of natural resources and this motivate our industries to develop existing and new processes, find a new use of waste and thus increase the overall efficiency. Food supply chain waste has become visible as a potential raw material for the production of fuels and chemicals globally. It contains a variety of different chemical components which can be utilized for many higher value applications. Biodiesel (ethyl ester) was prepared from waste cooking oil. Biodiesel fuel is becoming more demanding because of its environmental benefits as it is manufactured by natural resources such as plant, vegetable oil, and animal fats. Biodiesel contains higher cetane number than diesel fuel, it does not contains aromatics or sulfur and contains 10-11% oxygen by weight. Ethyl alcohol and sodium hydroxide used as a catalyst for the transesterification process.
Oil prices are increasing in international market so biodiesel could become a more viable alternative energy source. Oil feedstock is the major cost of biodiesel production. Hence if waste vegetable oil is used for biodiesel production, the economy of biodiesel will greatly improve and at the same time use of waste cooking oil also reduces the waste treatment costs.
Advancement in Heterogeneous Catalysis by the Application of Green Chemistry
Heterogeneous catalysis is an essential technology for developing benign processes in the chemical industry. Heterogeneous catalysis should offer excellent selectivity and high activity for the desired product and at the same time, it decreases the costs associated with product purification and waste disposal. In particular, an enhanced chemoselectivity is of primary importance in the processing of polyfunctional molecules. The irregular catalyst surfaces contain manifold active sites, which derive from a different other variety of undesirable reaction pathways. Recently surface modification has been clearly identified as an ideal technique for controlling the selectivity, enabling an intensified reactant orientation or fine-tuning of the active sites on the catalyst surface. Owing to their flexibility and versatility, organic modifiers represent an important set of compounds for the surface modification of noble metal nanoparticles. Although a variety of organic modification strategies have successfully improved the chemoselective reactivity, the application of this strategy has been limited to the confined catalyst field of metallic nanoparticles, especially noble metal nanoparticles. There is very less information is available on the modification of base-metal oxide catalyst with organic modifiers. It is a highly potential process of chemoselectivity redox chemistry because metal oxides are useful catalysts in the fields of chemistry bounded to redox properties with high stability and durability.
Reaction selectivity is defined by the diffusion, chemisorption, and surface reaction of the reactant in a Heterogeneously catalyzed reaction. These organic alteration techniques have enhanced nanoparticle catalysts by favorably influencing the diffusion or surface reaction step. There are many data that gives information the chemisorption behaviors of organic molecules despite that the effect of the organic modification on the reactant chemisorption affinity of a catalyst is not clear. This relationship remains important for the further development of this catalyst modification technique.
In quantitatively tuning the adsorbate affinity of the catalyst surface would strongly assist in determining the relationship between the reactant adsorption behavior and the reaction pathways.
Conference Highlights
- Green chemistry and catalysis
- Green Chemistry Metrics
- Green chemistry and Polymer technology
- Green catalysis in Petrochemical Industries
- Biocatalysis
- Green catalysis and Pollution control
- Green chemistry catalysis and fuel cell
- Green chemistry catalysis and Sustainable energy
- Green Chemistry in Pharmaceuticals
- Green Energy
- Green economy
- Green Synthesis designing
- New Trends in Green Chemistry
- Design of Next Generation Catalysis
- Nanotechnology and Green catalysis
To share your views and research, please click here to register for the Conference.
To Collaborate Scientific Professionals around the World
| Conference Date | November 15-16, 2018 | ||
| Sponsors & Exhibitors |
|
||
| Speaker Opportunity Closed | Day 1 | Day 2 | |
| Poster Opportunity Closed | Click Here to View | ||
Useful Links
Special Issues
All accepted abstracts will be published in respective Our International Journals.
- Organic Chemistry: Current Research
- Journal of Environmental Analytical Chemistry
- Chemical Sciences Journal
Abstracts will be provided with Digital Object Identifier by





































